

Background
In this second of three blogs on the work the ‘Dubbo’ group in Australia is doing we look at a controversial and rather difficult topic called interoception. Interoception deals with our awareness of our bodies internal state.
The field of interoception began to take off when it was found that small nerve fibers that feed sensory information from all over the body (on CO2 and oxygen levels, electrolyte salt levels, muscle metabolic by-products, pain, touch, etc.) converge in the dorsal horn of spinal cord.
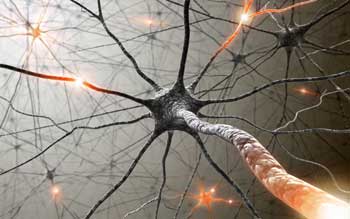
Small sensory fibers that merge at the spinal column and then shoot up to the brainsteam and insula provide the basis for the theory of interoception
From there the information loops into a main integration site in the brain stem which automatically maintains our breathing, metabolism, energy uptake and electrolyte balance. From the brainstem it move up into the anterior cingulate, insula and thalamus in the brain which, in turn, reach down and regulate activity in the brainstem. (A lesion in these areas will throw off regulation of breathing, cardiovascular functioning, etc.)
We should note that the interoceptive process is tightly tied to the autonomic nervous system. Those nerve fibers merge with the nerves leading from the vagus nerve to convey information first to autonomic nervous system centers that regulate homeostasis, and then to organs in the brain.
Once those signals get to the higher brain regions the brain is believed to form a picture of the physiological status of the body. If that picture indicates something is off in the bodies homeostasis the brain tweaks the autonomic nervous system and produces the emotions and behaviors needed to bring the body back into balance. The insula is essentially a place where a problem in the body shows up as an alarm in our consciousness. These Australian researchers believe the insula is where the alarm has gone off in ME/CFS.
The Study
This study examined whether an infectious event caused people who’d come down chronic fatigue syndrome (post-infective fatigue syndrome) to develop a) an overcharged autonomic nervous system and b) and an increased sensitivity to their body sensations – something they called ‘physiological hypervigilance’. They also examined autonomic nervous system variables during a cognitive test.
They determined the level of interoception present by measuring the participants awareness of their heartbeat and the point at which pressure on their skin induced pain. Heartbeat awareness has been used as an index of interoceptive sensitivity for years. Various measures of symptom, functionality, psychological distress and disability were also taken.
Results
High Levels of Interoception Hurt….But Are Not Tied to Mood
They found that the patients who were more aware of their heartbeat and more sensitive to pain tended to be more fatigued, have more mental and physical symptoms and be less functional.
No association between personality traits and interoceptive sensitivity was found. The correlation of psychological distress scores, disability and negative affectivity with increased sensitivity to pain but not to heartbeat discrimination suggested that pain was more of a cause of psychological distress and disability than an increased awareness of body sensations.
Cytokine Imbalance – The Gateway to Interoceptive Issues?
These findings strengthen the evidence of a pivotal role of a balanced cytokine response in dealing with acute infections
Why would people with ME/CFS be more aware of their body sensations? The participants in this study came down with ME/CFS after an infection and these authors proposed that something early in the infection had caused an unending up regulation of what they called the ‘neurovisceral regulatory circuits’.

Severe symptoms early in an infection greatly increased the risk of coming down with chronic fatigue syndrome in one study
Several years ago these researchers found that severe symptoms and high levels of cytokines early in the acute stages of infection set the stage for ME/CFS. Severe symptoms early in the infection, in particular, played an extraordinarily strong role in determining who came down with chronic fatigue syndrome afterwards; people with severe symptoms were 200 x’s more likely to come down with ME/CFS than people with less severe symptoms.
The evidence therefore strongly suggests that events occurring early during the acute infection and linked to illness severity and the intensity of the inflammatory response are critical to understanding the development of PIFS.
The cytokine levels suggested an unbalanced immune system early in the infection played a key role in who came down with chronic fatigue syndrome. Since they were unable to find evidence of increased cytokine or pathogen levels later in the they suggested that the die is cast, so to speak, very early in the illness- at least for pathogen triggered ME/CFS patients.
Genetic Basis
Next they looked to see if a genetic basis for an unbalanced immune response was present. They knew that slightly altered forms of genes (called polymorphisms) have been associated with greater susceptibility to or worsened outcomes of several infections including Epstein-Barr virus, tuberculosis, sepsis, malaria, etc. but no one had looked at ME/CFS.
They found that these unsuspecting, soon to be chronically ill patients indeed carried altered forms of two immune genes for IFN-y and IL-10 genes that could set the stage for an unusually strong, unbalanced immune response
Their IFN-y gene suggested that when met with an infection, they would pump out high levels of IFN-y- a pattern that’s associated with more severe cases of tuberculosis and may play a key role in infectious mononucleosis – a common way to get ME/CFS.
Couple that with a tendency to produce less IL-10, a proinflammatory cytokine inhibitor, and you may have set the stage for an overheated immune response to infection that fried some important ‘neurovisceral circuits’. Individuals with these polymorphisms were eight times more likely to come down with ME/CFS than those who didn’t have them.
Cell cultures indicated the cells of these individuals were pouring out the pro-inflammatory cytokines IL-1B, TNF-a, IL-6 as well. Each of these proinflammatory cytokines has been associated with ME/CFS at one time or the other. They have been associated with pain hypersensitivity (IL-1B) and fever and inflammation (TNF-a, IL-6).
Pathway to the Brain
We suspect that in individuals with a prolonged postinfective illness, a prominent cytokine response in the acute phase causes sensitization in these CNS pathways
From the beginning of the Dubbo studies, the lead researcher, Dr. Andrew Lloyd pointed to the brain. In the gene polymorphism paper the Dubbo researchers pointed to the remarkable fact that a single exposure to bacterial (LPS) triggered TNF-a in the brain can send the immune cells in the brain – the microglia – into a tizzy for up to a year.
They’re not alone in thinking that immune activation can tweak with the way the brain views the body. Vaccinations have been associated with activation of the brain regions believed involved in interoception. Indeed, ‘ interoceptionists’ count the symptoms that show up during an infection (‘sickness behavior’) as an interoceptive process.
It is plausible that in vulnerable individuals a severe immunological stressor leads to sensitization in interoceptive pathways which manifests as physiological hyper-vigilance resulting in the symptom complex characteristic of post-infectious fatigue.
An Autonomic Hypervigilance
Since they published this paper this group has documented autonomic dysfunction in their post-infective ME/CFS patients. Autonomic abnormalities have been associated with trouble ‘learning’ in both ME/CFS and FM patients. The Vollmer-Conna/Lloyd group found that the heart rates of healthy controls dropped during a cognitive test, but that the heart rates of post-infective fatigue patients actually continued to increase. This pointed to an autonomic hypervigilance that fit very well, the authors believed, with the pattern of increased interoception in this study.
The authors suggested that these sensitized circuits had lead ME/CFS patients to ‘an internal focus of attention on the(ir) disease state” which could lead to the interpretation of any physiological disturbance as being ‘pathological’. In fact, they suggested that enormous fatigue many people with ME/CFS feel after exertion could derive from this process.
Interpretation
The authors went a long way when they suggested that post-exertional malaise might be due to an internal focus of attention on one’s ‘disease state’. That surprised me given theirs and others findings of autonomic problems. They did note that the autonomic hypervigilance they and others have found in ME/CFS fit with their interoception theory. Indeed, a recent study suggested that the same parasympathetic withdrawal/sympathetic dominance found in ME/CFS/FM is associated with high levels of interoception.
To conclude that post-exertional malaise is simply the result of a heightened awareness of ones internal state, however, is to ignore evidence that something physiologically often goes awry in ME/CFS/FM during exertion. As evidence of muscle and ANS problems accumulates in both ME/CFS and FM a brain focused, top-down interpretation of the increased pain/fatigue found in both is losing some of the traction it’s had. (Look forward to a fascinating blog on this issue in FM coming up :)).
There’s no reason both body and brain problems aren’t both occurring. For me, I’m content to consider the possibility that something heightened my sensitivity to body sensations; I have too many of those to ignore that possibility, and other research suggests that the proposed seat of interoception, the insula, is activated in ME/CFS and FM. I’m also very comfortable with Gupta’s idea that ‘watching my body’, ie attention to my body symptoms is not a good idea.
(The insula is involved in more than body awareness, though. Another study suggested the insula played a key role in the hearts response to stress. Another that it helped produced the same kind of parasympathetic downturn/sympathetic upturn in tinnitus that’s seen in ME/CFS.)
It’s still somewhat incredible to me and my exertionally challenged state, though, how little impact the Pacific Foundations (now Workwell Foundation) exercise studies have had on the prevailing interpretations of this disorder. Yes, exercise is commonly used in research studies, and yes, some studies have validated their finding but Workwell’s central finding that a unique metabolic problem is present a significant number of ME/CFS patients has never assumed a fundamental position in this field.
It’ll be fascinating to see the results of Klimas’ teams study indicating that autonomic nervous system problems kick off an immune cascade that ultimately results in PEM and the Lights continuing research suggesting that sensory data from over-active sensory receptors is slamming the central nervous system. (The Vollmer-Conna/Lloyd group has been unable to validate their findings but more is on the way from them.)
- Nervous System Abnormalities Tied to Cognitive Problems in Chronic Fatigue Syndrome – Part I of the Australian Research Series
- Next up we’ll see the Australian team integrate their autonomic nervous system findings into their brain research to come up with a new model of ME/CFS.







Cort,
I’m very happy to watch my body closely for signs that I might need to stop, sit or lie down. If I’m out of breath, or even puffing, I know I am over my anaerobic threshold.
I also get very subtle signals that I need to stop what I’m doing, and lie down or change to something else. Occasionally slight toxic tastes in my mouth suggest the same thing. These minute symptoms if attended to mean that very brief flat eyes-closed rests can bring me refreshment and further energy.
We may still be singing from the same hymn book here. Other than physical fatigue, I have a pretty good handle on social/emotional cognitive and environmental fatigue problems with my moderate CFS/ME. In other words what I am listening to is not CFS/ME symptoms but my body’s subtle signs or change. Acting on these (pacing and flat rests) seems to be very helpful to me.
I wonder if any others of your readers find the same?
I’m of two minds. Too much watching isn’t good for me; I find it best to be engaged in what I’m doing as much as I can. On the other hand ignoring symptoms can be a recipe for disaster as well as I’ve proved many times over the years..
Actually it’s still really hard for me to know when to push and when not to….I pushed the other day and did fine…then I felt pretty good and did some physical work and now I’m basically huddled in pain – my muscles feeling like they’re on fire. I think this is probably because I’ve been doing too much over the past couple of weeks but it’s still pretty much a mystery to me (lol)…
I think they’re missing something with interoception. I just can’t imagine that this whacky response to exertion is the result of an overheated insula misperceiving the body; it’s just too painful for one thing and the more I exercise the more pain increases. I think they’re onto something with the ANS, on the other hand.
The only thing I can conclude is that the body pains and symptoms are pretty much always there and there are alot of them.
There were only 23 people in the sample, and, from the graphs that I saw, the results greatly overlapped the results of the healthy group. I haven’t been able to read the full study, but this was a small scale statistical exercise, so it wouldn’t be worth reading anything much into it. Unless the sample was truly random (which is next to impossible in these sorts of tests), the results cannot be taken seriously.
(Just to spell it out, people with ME who are likely to volunteer for studies on ME are more likely to be troubled by their symptoms: those who have been able to come to terms with their symptoms, or whose symptoms are less severe, are more likely to be getting on with things. It’s not rocket science.)
I’m thinking of setting up a study to find out if people with ME are more likely to volunteer to take part in studies of ME than healthy people. Of course I will need funding, and I will need to find some volunteers to take part. Any offers?
Thanks for pointing that out Graham…Another small study…..I have the feeling that we need some hundred person studies with all there heterogeneity in this illness.
Hi Cort,
I think you’re on the right track, but it seems to me a mistake to keep reflexively blaming these kind of phenomena on “mystery virus” when there are environmental biotoxins that are known to create all the effects that you are considering here.
You’ve put enough thought into these issues that I think it will be clear why this article might be relevant.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2525487/
Of course, a question here is how it could be that enough of a toxin similar to this stuff could be present in the environment for people to be being made sick by it with these symptoms.
That would be pretty scary if it were the case, wouldn’t it?
If you have time to read this paper, let me know your thoughts. Thanks for your article.
Best, Lisa
That’s just perverse. In fact, in my case I had no idea that I had tachycardia until it was picked up in a routine a visit at 130 bpm. The doc was like don’t you notice this?
A very interesting mode of action Lisa :
“DOM is structurally similar to another known toxin, kainic acid [KA].
Both are excitatory amino acid [EAA] analogues of glutamate, a major excitatory neurotransmitter in the brain that is known to activate glutamate receptors [GluRs].
DOM induces excitotoxicity by an integrative action on ionotropic GluRs [iGluRs] at both sides of the synapse for which it has high affinity, preferentially the KA subtype, coupled with an effect that prevents the channel from rapid desensitization [48,59–79].
A synergistic effect of DOM with endogenous glutamate and NMDA receptors agonists, has been demonstrated in vitro and in vivo, which contributes to the excitotoxicity [67,79,80].”
In other circles, paying attention to symptoms is called biofeedback (this can be done with fancy equipment, but if I’m not mistaken it can be done on own, too; when I had a formal session with equipment, I was already self-correcting to best available and they could do very little for me, to their surprise). This is deemed a positive coping mechanism and is done to minimize symptoms via whatever avenues are available (adjusting position, taking breaks, hydrating, etc.).
I suspect if they are finding increased introspection and if it were not an artifact of small sample size, it would be due to having a disease with no specific treatments available (at least not to the vast majority of patients; some have success with antivirals, etc., but this is not often available) and often having either minimal or no symptomatic treatments offered. Thus, patients perform biofeedback on their own as a way of self-managment. Not with great success, but better than not trying.
(This is not the same as focusing on symptoms in a worrying or moping sort of a way; it is simply noticing what is going on, moderating whatever is in one’s power to moderate, and accepting the rest for the time being – obviously we hope not to have to feel like this forever)
all the best,
Janelle
Given what they believe, biofeedback is the next natural step for this group and they are actually either in the midst of a biofeedback study or have completed one. Dr. Vollmer-Conna told me that some of the participants did very well on biofeedback and some got worse.
I think I’m probably doing biofeedback quite a bit. When I notice my breath rising and my muscles contracting and heart beginning to pound I try to shift into a calmer state…
Are people with CFS more receptive to pleasurable sensations?
I don’t know about ‘receptive’ but a pleasurable activity can knock me off my feet just as easily as a ‘stressful’ one.
Any overstimulation does it whether negative or positive.
I have the same reaction…Getting really excited about something tends to ‘lock me up’ as well….
Instead of an infection couldn’t your own immune system trigger this, or even high levels of stress hormones? This would explain the small percentage that come down with CFS without an infectious trigger. This sounds a similar mechanism as Autism and could be very possible.
I would think so – the immune activation is the key – with a genetic predilection to a strong immune response to help out.
I do not believe the aussie researchers theory. These researchers think that ME is a psychiatric illness. There wrong. It is a neurlogic problem. It is not normal if the brain and immune system respond abnormal to all kinds of stimuli. Because that is the key problem.
I’ve no problem with the idea of altered levels of sensitivity (e.g. as a result of inflammation), but to suggest this is the cause of CFS symptoms, is naive, in my opinion.
If we think about what it’s like to have flu, for example, the symptoms can be similar to ME. But we don’t say that flu is caused or perpetuated by an increased sensitivity. I can have altered states on consciousness when I have flu… I can be semi-conscious, or agitated, or I can have an enhanced sensory perception, or I can experience a pleasant state of calm (sitting under the duvet, watching TV, drinking lemon and honey, and feeling blissfully at ease with the world, while I relax and give myself permission to be at one with my duvet.)
But these changes are all a result of an immune response, and not a cause of the illness or symptoms.
As Cort suggests, research is moving beyond this kind of theory where the central nervous system (i.e. ‘top down’) is said to be responsible for all symptoms.
In other words, if there is increased (nervous-system) sensitivity in ME/CFS, I believe that the sensitivity is a symptom, and not a cause of symptoms.
(A symptom caused by inflammation or an immune system issue.)
http://www.ncbi.nlm.nih.gov/pubmed/20882373
Curr Rheumatol Rep. 2010 Dec;12(6):448-54. doi: 10.1007/s11926-010-0134-x.
Is it all central sensitization? Role of peripheral tissue nociception in chronic musculoskeletal pain. Staud R.
Abstract
Fibromyalgia syndrome (FM) is a highly prevalent musculoskeletal disorder that is often accompanied by somatic hyperalgesia (enhanced pain from noxious stimuli). Neural mechanisms of somatic hyperalgesia have been analyzed via quantitative sensory testing of FM patients. Results of these studies suggest that FM pain is associated with widespread primary and secondary cutaneous hyperalgesia, which are dynamically maintained by tonic impulse input from deep tissues and likely by brain-to-spinal cord facilitation. Enhanced somatic pains are accompanied by mechanical hyperalgesia and allodynia in FM patients as compared with healthy controls.
FM pain is likely to be at least partially maintained by peripheral impulse input from deep tissues. This conclusion is supported by results of several studies showing that injection of local anesthetics into painful muscles normalizes somatic hyperalgesia in FM patients.
I’m of a similar mind Cort. Watching my health to closely can led to way to much self reflexion this in turn
broadens my symptom range, but I’m not sure this doesn’t happen with other illnesses, especially autoimmune, like RA or MS. If this boils down to controlling ones thoughts/emotions in order to achieve a type of “wellness” then I think it’s way off. The onset of some forms of ME/CFS may have something to do with emotional trauma and some mix of pathogen.
But we should be careful not to link the illness with brain retraining. This not like an obesity, diabetes link.
I’ll bet that ‘de-stressing’ is used in many cardiovascular disorders and I’ll be that because of the autonomic component in the cardiovascular system and the fact that it can be controlled somewhat.
Check this out from an abstract – http://www.ncbi.nlm.nih.gov/pubmed/23184602
Psychological distress can trigger acute coronary syndromes and sudden cardiac death in vulnerable patients. The primary pathophysiological mechanism that plays a role in stress-induced cardiac events involves the autonomic nervous system, particularly disproportional sympathetic activation and parasympathetic withdrawal. This article describes the relation between psychological distress and autonomic nervous system function, with a focus on subsequent adverse cardiovascular outcomes.
Results of the systematic review indicate that the primary brain areas involved in the autonomic component of the brain-heart association are the insula, medial prefrontal cortex, and cerebellum (based on 121 participants across three studies that fitted the inclusion criteria). Other areas involved in stress-induced autonomic modulation are the (anterior) cingulate cortex, parietal cortex, somatomotor cortex/precentral gyrus, and temporal cortex. The interaction between central and autonomic nervous system responses may have implications for further investigations of the brain-heart associations and mechanisms by which acute and chronic psychological distress increase the risk of myocardial infarction, cardiac arrhythmias, and sudden cardiac death.
That’s why I say de-stress as much as possible and don’t worry about what it ‘means’. ME/CFS is a disorder of the autonomic nervous system (stress response system) and immune system and probably others. We know that blood pressure, blood volume, heart rate, etc is messed up in ME/CFS. We know the same kind of ANS ‘signature’ applies to it and heart disorders….Given that is there any wonder that ‘stress’ can be hurtful? Or that becoming experts in stress management might be a very good thing to do? Hey, the two biggest stress response systems in the body are involved…
I feel like I have to say that this is seriously lousy “research.” When will this stop… why is this crap published?
The primary outcome measures are subjective… and their conclusions of those measures do not make sense given the differences in the groups. They clearly stated that between the normal group and the PIFS groups that PIFS had significantly higher baseline heart rate. You cannot draw any conclusions from a group’s perception of their heart beats when the two groups have significantly different baseline rates. Seriously? Anyone knows its easier to tell when your heart is beating if there is increased chronotropy and I would be willing to bet a lot that if ECHO’d the PIFS group would have significantly higher inotropy compared to normals as well making it even easier to tell.
In my opinion the so called “autonomic dysfunction” is greatly overstated in ME. We have what appears to be abnormal autonomic function to be sure (I for one have pretty bad POTS), but if that was the major problem then it would be fairly easy to make people MUCH better if not cured as we have plenty of drugs that can change autonomic function as desired. The autonomic “abnormalities” found in ME are what we would consider a normal response to low blood volume, cardiac dysfunction, and some metabolic diseases – all things that we already know are abnormal. I wish researchers would not pick abnormalities to study when the “abnormalities” are really just compensatory mechanisms for deeper problems.
If you are right then the ANS overactivity is a compensatory mechanisme. This is a very important and critical issue. But we mus know this for sure. I think you are right.
I wondered about that as well but all studies that I’ve read have indicated decreased contractility, decreased stroke volume, etc in ME/CFS. I’ve never seen anything that suggests cardiac output is increased. The only thing that’s increased is heart rate and my understanding its the force of the heart beat not the heart rate that’s determinative in these tests.
Am J Med Sci. 2003 Aug;326(2):55-60.
Abnormal impedance cardiography predicts symptom severity in chronic fatigue syndrome.
Peckerman A, LaManca JJ, Dahl KA, Chemitiganti R, Qureishi B, Natelson BH.
RESULTS:
The patients with severe CFS had significantly lower stroke volume and cardiac output than the controls and less ill patients. Postexertional fatigue and flu-like symptoms of infection differentiated the patients with severe CFS from those with less severe CFS (88.5% concordance) and were predictive (R2 = 0.46, P < 0.0002) of lower cardiac output. In contrast, neuropsychiatric symptoms showed no specific association with cardiac output. Psychosom Med. 2001 Sep-Oct;63(5):756-64.
Cardiovascular responses of women with chronic fatigue syndrome to stressful cognitive testing before and after strenuous exercise.
LaManca JJ, Peckerman A, Sisto SA, DeLuca J, Cook S, Natelson BH.
.
METHOD:
Beat-by-beat blood pressure and electrocardiogram were recorded on 19 women with CFS and 20 healthy nonexercising (ie, sedentary) women while they performed cognitive tests before, immediately after, and 24 hours after incremental exercise to exhaustion.
RESULTS:
Diminished heart rate (p <.01) and systolic (p <.01) and diastolic (p <.01) blood pressure responses to stressful cognitive testing were seen in patients with CFS when compared with healthy, sedentary controls. This diminished stress response was seen consistently in patients with CFS across three separate cognitive testing sessions. Also, significant negative correlations between self-ratings of CFS symptom severity and cardiovascular responses were seen (r = -0.62, p <.01). CONCLUSIONS: Women with CFS have a diminished cardiovascular response to cognitive stress; however, exercise did not magnify this effect. Also, the data showed that the patients with the lowest cardiovascular reactivity had the highest ratings of CFS symptom severity, which suggests that the individual response of the patient with CFS to stress plays a role in the common complaint of symptoms worsening after stress. Clin Physiol. 1999 Mar;19(2):111-20.
Cardiovascular response during head-up tilt in chronic fatigue syndrome.
LaManca JJ, Peckerman A, Walker J, Kesil W, Cook S, Taylor A, Natelson BH.
This study examined the cardiovascular response to orthostatic challenge, and incidence and mechanisms of neurally mediated hypotension in chronic fatigue syndrome (CFS) during a head-up tilt test.
Among subjects who completed the test, those with CFS had higher heart rate and smaller stroke volume (P < 0.05) than corresponding control subjects. When comparing those who had a positive test outcome in each group, CFS patients had higher heart rates and lower pulse pressure and pulsatile-systolic areas during the last 4 min before being returned to supine (P < 0.05). These data show that there are baseline differences in the cardiovascular profiles of CFS patients when compared with control subjects and that this profile is maintained during head-up tilt. However, the frequency of positive tilts and the haemodynamic adjustments made to this orthostatic challenge are not different between groups. I agree that the autonomic dysfunction may very well be compensatory.
Your idea made me look further and find a study I had missed. Check this out – more severe people with CFS had lower cardiac volume but then they corrected for that they still had reduced contractility – so that issue was separate (:))
Clin Sci (Lond). 2009 Oct 19;118(2):125-35. doi: 10.1042/CS20090055.
Chronic fatigue syndrome: illness severity, sedentary lifestyle, blood volume and evidence of diminished cardiac function. Hurwitz BE, Coryell VT, Parker M, Martin P, Laperriere A, Klimas NG, Sfakianakis GN, Bilsker MS.
The study examined whether deficits in cardiac output and blood volume in a CFS (chronic fatigue syndrome) cohort were present and linked to illness severity and sedentary lifestyle. Follow-up analyses assessed whether differences in cardiac output levels between CFS and control groups were corrected by controlling for cardiac contractility and TBV (total blood volume).
Controls were matched to CFS participants using age, gender, ethnicity and body mass. Echocardiographic measures indicated that the severe CFS participants had 10.2% lower cardiac volume (i.e. stroke index and end-diastolic volume) and 25.1% lower contractility (velocity of circumferential shortening corrected by heart rate) than the control groups. Dual tag blood volume assessments indicated that the CFS groups had lower TBV, PV (plasma volume) and RBCV (red blood cell volume) than control groups.
Lower cardiac volume levels in CFS were substantially corrected by controlling for prevailing TBV deficits, but were not affected by controlling for cardiac contractility levels. Analyses indicated that the TBV deficit explained 91-94% of the group differences in cardiac volume indices.
Therefore the findings indicate that lower cardiac volume levels, displayed primarily by subjects with severe CFS, were not linked to diminished cardiac contractility levels, but were probably a consequence of a co-morbid hypovolaemic condition. Further study is needed to address the extent to which the cardiac and blood volume alterations in CFS have physiological and clinical significance.
I am sorry I was not more clear…
First, I was simply making the point that given the populations this study used and their endpoints that the conclusions they make are not logically coherent (i.e. this is bad science).
Second, I believe the increased heart rate and EF (sympathetic activation) occurs BECAUSE of the overall decreased cardiac function (cardiac output or index) as you have noted. That is the normal physiologic response to lowered cardiac output… it happens to people that are in heart failure all the time. I was in no way suggesting that cardiac output is elevated in ME patients… IF it was elevated that would go along with the theory that our disease is “mainly autonomic dysfunction” (that is why I think blaming primary autonomic dysfunction when you have low cardiac output at the same time does not make sense).
Unfortunately there are very few studies that even look at cardiac physiology in ME in any sort of detail – you got most of the ones I know about above. The only person I know of who is actively looking at cardiac dysfunction on a regular basis is Dr. Cheney. Hopefully he will publish his work or it will be replicated by someone else and published. Unfortunately, the way things are right now is that most cardiologists would totally miss most of the abnormalities we have simply because they do not look for them: a standard ECHO does not measure cardiac output/index and most parameters that look for diastolic dysfunction.
Here are a couple from Hollingsworth you didn’t mention – they are small but well done:
Impaired cardiovascular response to standing in Chronic Fatigue Syndrome
and
Impaired cardiac function in chronic fatigue syndrome measured using magnetic resonance cardiac tagging
by
Kieren G. Hollingsworth, et al.
The following is a review article regarding “normal” heart failure and mitochondrial dysfunction… Is not a ME article, but worth a read considering the cardiac issues in this disease are almost certainly energy related.
The Failing Heart — An Engine Out of Fuel
Stefan Neubauer, M.D., F.R.C.P.
N Engl J Med 2007
I live in work in a health-research related field in the same city as Dr Lloyd (Sydney) and have followed his ME/CFS research activities for many years. To my mind, his Dubbo study is a good example of fact-based science, where, as you may know, approx. 10% of people coming down with severe viruses and other pathogens developed post-infectious fatigue after 6 months. However, I have been astounded to see him recently announce categorically in the national media that laboratory-based abnormalities will not be found in ME/CFS! That is speculation at best and psychiatric prejudice at worst. This is despite the clear findings of immune-system abnormalities in ME/CFS coming out of the National Centre for Neuroimmunology and Emerging Diseases (Australia), led by Professor Sonya Marshall-Gradisnik, as they develop a set of potential biomarkers for ME. For that see: http://www.theaustralian.com.au/news/breaking-news/aust-bid-to-solve-chronic-fatigue-riddle/story-fn3dxiwe-1226643151384
Wow..Are you sure of that? They’ve certainly found abnormal heart rate variability in ME/CFS and as you note NK cell dysfunction is pretty and we’re getting more brain findings.
Hi Cort,
Dr Andrew Lloyd on Australian ABC Radio “Life Matters” (5th March 2012) discussed CBT for CFS, referring to the the Dutch Study of teenagers using online CBT. The audio can be heard here:
http://www.abc.net.au/radionational/programs/lifematters/web-based-therapy-for-cfs/3865870
Here is my summary of Lloyd’s views as presented in this program:
Lloyd believes that a virus hits the patient then the virus goes away but leaves behind lasting damage to the brain (the “viral hit and run theory ” of CFS). He thinks CFS is started by a range of different infections that cause imbalance then CFS is maintained by a number of secondary factors, in particular:
– messed up sleep/wake cycle
– feeling depressed and down
– exercise avoidance / fear of exercise
He thinks that CFS is a disorder of the brain: not in the muscles or the heart and not caused by a pathogen sticking around. For example, when blood samples were taken from people who got Glandular Fever over a 12 month period, the persistence of the virus is no different between those who went on to get CFS and those who didn’t get CFS.
He says that there is clear evidence that various infections triggers CFS, and there is no evidence that certain personality types or socioeconomic classes are more prone to CFS.
Then he says:
– CFS cannot be equated with a mood disorder such as depression or anxiety.
– There is a mild female predominance, slightly more women than men get CFS.
– CFS peaks at 40 yrs of age (most common there).
– People get CFS for weeks, months and for “some people, years”.
– Teens and young people recover in greater numbers than older people who get CFS, probably due to psychological factors (he didn’t present any evidence for this conjecture). He speculates that when you get the teens back to school then they sort of “click” mentally and their health starts to improve.
The evidence for CBT being helpful for CFS is very strong, but CBT is not curative, it can help patients manage their day and reduce symptom severity so that then “the natural history of resolution kicks in” and they can get gradually better.
Dr Lloyd is also quoted elsewhere as saying that, “If you had to choose a place in the body where the problem lies we’re already 99.9% confident the problem is in the brain and we already have some ideas about how the brain might be disturbed to generate this illness”, but the journalist may have attributed what Dr Hickie said to Dr Lloyd. See the report at: http://theconversation.com/chronic-fatigue-focus-shifts-from-viruses-to-the-brain-9713
I haven’t been able to find his comment about immunological testing being useless in CFS.
Ian thanks so much for that great summary!
Cort, you mentioned a third blog.
—–
In this second of three blogs on the work the ‘Dubbo’ group in Australia is doing
—–
The only other one I could find was: http://www.cortjohnson.org/blog/2013/05/24/nervous-system-abnormalities-tied-to-cognitive-problems-in-chronic-fatigue-syndrome/