



“It really provides a whole new tool to our toolkit.” Philip Gehrman, Ph.D., CBSM
Sleep or the lack of it is a serious issue in chronic fatigue syndrome and fibromyalgia. Not just your energy level but your ability to think clearly, your sensitivity to pain and your emotional well-being can be affected by the amount and depth of sleep you get.
Despite the various sleep medications present many are not getting deep, enriching sleep and insomnia remains a problem. A new sleep medication with a novel mechanism due out later this year could change that.
It fact it, and the drugs expected to follow it, could create a sea-change in how sleep issues are treated.
It all started about 15 years ago when two research teams stumbled upon a new receptor. Further research lead to a fundamental re-assessment of how sleep happens (or does not happen) and, ultimately the development of a new class of sleep drug.
The neurons that promote wakefulness (called orexin neurons) do not get turned down at night in insomniacs. Suvorexant reduces orexin neuron activity, hopefully allowing insomniacs to go to sleep.
Insomniacs on orexin fell asleep more quickly than those on placebo, and slept longer, and had fewer awakenings during sleep in the latest clinical trial. Animal studies suggest the drug affects cognitive functioning less than current sleep drugs. The fact that all stages of sleep improved suggested the drug might have broad applications in ME/CFS where different types of sleep disturbance occur. Results can occur as quickly as the first night.
FDA Wariness
Nine clinical trials (and three phase III clinical trials) later, almost everyone is betting the FDA will approve its first new insomnia drug in over thirty years this year. In a review analysts called ‘tough’, however, the FDA expressed wariness about the daytime sleepiness some participants experienced at higher doses. After halving the dosage for Ambien in women because of sleep-walking and other issues, the FDA is keeping a close watch on sleep inducing drugs.
The potentially large number of people – from young to old, some on other drugs – taking a new drug with a new mode of action raises concerns at the FDA.
After hundreds of reports of car accidents due to Ambien, the FDA is now requiring driving tests to assess the impact sleeping drugs have on driver safety. Several participants on higher doses who swerved out of their lane too often suggested suvorexant could pose a problem for drivers the next morning.
After noting that suvorexant stays in the body 8 times longer than Ambien, and that people on the drug cannot assess their driving ability accurately, Public Citizen spoke against its approval.
The FDA panel, however, voted 10-3 that suvorexant is safe at 15/20 mgs, and 8-7 that it is not safe at 30/40 mgs. The FDA is expected to approve the drug at lower doses.
From the Hammer to the Scalpel A New Approach to Enhancing Sleep
One of the most exciting things about suvorexant is how different it is. According to Dr. Daryle Shoepp of Merck Labs, most sleep drugs work by turning off GABA neurotransmitters, but because they turn off GABA receptors all over the brain they essentially bludgeon the brain to sleep.
Because they turn off GABA receptors indiscriminately Ambien-like drugs can be used to prevent convulsions, to reduce anxiety, etc., and help people to fall asleep. Benzodiazepines effectively enhance short-term sleep, and, in fact, may be more effective than suvorexant at short term sleep inducement, but problems with tolerance and dependence have left sleep specialists clamoring for better drugs. Withdrawal from benzodiazepines can even result in increased insomnia.
Developed to target a key process in sleep first identified in lab animals and then in humans, suvorexant is a scalpel to GABA drugs hammer. In contrast to the billions of neurons the current crop of sleep drugs affects, suvorexant targets about 100,000 neurons in the hypothalamus.
Studies suggest suvorexant has fewer side effects than benzodiazepines, and it is not addictive. A yearlong study suggested suvorexant had good staying power and produced few side effects with about 10% reporting morning drowsiness. Despite the availability of lower-cost alternatives analysts project sales could reach $900 million dollars in the first year.
More Applications on the Way (?)
Orexin neurons in the LHA are anatomically well placed to provide a link between the limbic system, energy homeostasis, and brain stem monoaminergic or cholinergic neurons….
These findings indicate a critical role for orexin neurons in the regulation of vigilance states, according to internal and external environments, for survival. Yamanaka and Inutsuka
Do not be surprised if suvorexant does not end up affecting more than sleep. Concentrated in the hypothalamus, orexin neurons axons extend throughout the brain.
Autonomic Nervous System – The ‘always on’ orexin neuron issue, interestingly, appears to resemble a similar autonomic nervous system (ANS) issue in ME/CFS. Programmed to always be ‘on’, the sympathetic nervous system (SNS) is only turned off when the parasympathetic nervous system (PNS) tells it to calm down. The default position, for the ANS, in other words, is to have the SNS ‘on’ so that it can quickly respond to an emergency situation that requires that we fight or flee.
The inability of the parasympathetic nervous system, including the vagus nerve, is to turn the SNS off, appears to contribute to the hypervigilance and ‘wired but tired’ feeling found in ME/CFS.
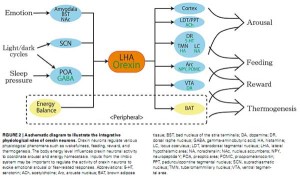
Orexin neurons extend throughout the brain and affect many functions including the autonomic nervous system
Given that, orexin regulation of the ANS is intriguing. Orexin’s stimulation of the sympathetic nervous system suggests that orexin inhibitors such as suvorexant might be able to help reduce the ‘arousal’ present in ME/CFS. Propranolol’s ability to reduce orexin induced heart rate increases suggests it could be impacting that system in ME/CFS.
Knockout mice without orexin neuron functioning demonstrated reduced cardiovascular responses to an emotional stress, another issue with ANS implications, which may come to bear in ME/CFS.
Cerebral Glucose – With the evidence of reduced cerebral glucose concentrations in ME/CFS, it is intriguing that reduced glucose triggers orexin neuron activity (and increased wakefulness). Orexin’s stimulation of the histaminergic neurons of the brain is intriguing given the interest in histamine’s role in ME/CFS.
Glutamate – Then there’s the fact that glutamate, a possible player in ME/CFS and other neuroinflammatory disorders, activates orexin neurons.
Merck is also tinkering with a similar ‘molecule’ they believe may be able to affect migraines, neuropathic pain and depression.
Conclusion
Suvorexant’s expected FDA approval will bring a new class of sleep drug to ME/CFS/FM patients suffering from insomnia. Targeting a sleep center of the brain, suvorexant may be a better fit for chronic insomniacs than the anti-hypnotics available now. The drugs ability to down regulate sympathetic nervous system functioning suggests it could have ancillary benefits as well.








CORT- AGAIN- I SAY MOST OF THE PEOPLE WITH ANY OF THE SLEEP PROBLEMS-NEED TO SEE A SLEEP SPECIALIST AND TRY A CPAC MASK. THIS HAS HELPED ME TREMENDOUSLY. WE ARE ALL OVERMEDICATED ALREADY.
SAN DIEGO#1
I can’t wait to see if there is a medication out that will help with CFS/ME/FMS. I use a Cpac machine (6 yrs) it does not make any difference in the way I feel during the day time. My strength is terrible. I’m only 65 and should be able to do much more than what I can do. If I over do I pay for it at least 3 days after. That don’t give me much time to be able to enjoy a one week vacation. If I could find one medication that would work for the ones I take now, then I would be taking less meds.
I would be very happy then,
Kathytweety
We’re a heterogeneous group. What works great for one person has no affect in another. I think we can expect a wide range of responses with regard to everything…Which means you really have to try just about everything to know….unfortunately (lol)
For me vacations are tough because its so hard to pace yourself…
Dear Cort, well, this is exciting for me. I so relate to the “bludgeoning the brain to sleep”, as that is what I have to do and I know I am hyper-vigilant, even though I no longer need to be. Right now I am taking three medications to sleep, not counting melatonin, L-theanine, etc. I will do anything to get sleep and that is because I have not awakened rested in 30 years. Even with the Ambien etc. I wake often and have to re-medicate most nights. I noticed the docs trying to crunch me out of my meds, I now see because of this research on Ambien, but I get so stressed when I consider what my nights would be like without their aid. I would love to try this new drug. Do you know when it will be available? Gratitude to you for this blog. warmly, joya
The FDA could make a decision anytime and will certainly do so before the end of the year.
I read somewhere that emergency room visits due to Ambien increased some ungodly amount over the past couple of years. That really alerted the FDA to some issues…
I didn’t realize that these hypnotics basically shut down GABA receptors across the entire brain (!). Hopefully a targeted approach will work better and there will be more targeted approaches in the future.
And then, too, perhaps the FDA needs to realize that some of us are so disabled by our illness that we do very little driving—we hardly leave the house. I hope physicians are allowed to prescribe what their patients need and what works. I have not reduced by dose of Ambien (I’m a woman!!) but I also don’t drive when I get up in the morning!!! On the brighter side, I am excited that a new drug is coming because I have no doubt that the Ambien is helpful but I still sleep poorly. I think if we all slept better, we’d feel better!!! Please FDA we want help!!
Hi again, Cort. I have heard some stories too. But I never take my Ambien until I am nearly ready to turn the light out. And I don’t take much at a time and it lasts me only 1 1/2 – 2 hours. A longer half life would be good for me. I would be more concerned if I wasn’t so desperate. I have heard one of my musician friends that took his Ambien after a gig, before getting to the motel and left a big piece of his car at the gig site. I just thought that was a bit stupid, but then he was drinking too. Of course, I would rather just get well. Getting out of this 8000 ft. altitude is going to help a lot as soon as I can muster the energy to pack! haha. (not funny) joya
I do agree that many may have apnea, as this is also a problem that I have. I do use a CPAP and it does seem to improve my sleep. There are two types of apnea one is obstructive in the throat where the muscles relax and close in on itself (this can happen, no matter what your weight.), and also central sleep apnea where your brain doesn’t tell you to breathe. If you have central sleep apnea and you take a sleeping pill there is the potential for you to stop breathing any of the time and well we know what that would bring . . . .you wouldn’t have a sleep problem —you’d have eternal sleep. 🙂 So, I agree, that if there is a sleep problem (and there usually is with any of these autonomic problems) you need to first find out if either of these two things could be an issue. We for sure need oxygen and if we are waking ourselves up to make ourselves breathe –no doubt there isn’t restful sleep.
Then with this med. if it sticks around in your body for so long – seems this could make CFS/ME worse – as you would feel more tired. It also makes me question whether or not it plays a role with acetylcholine and would it affect brain function and cause memory problems. I think this is too new to experiment with. I’ll let someone else be the guinea pig on this one. Thanks for the info, Cort.
Issie
It does stick around longer than some other drugs which is a concern I would think if some people are not detoxing drugs well.
On the other hand, next day sleepiness, I think I forgot to mention in the blog, was present in about 10% of those taking the drug and 90% didn’t have that problem so that may or may not be an issue, depending on how you respond. In regular insomniac’s it had less cognitive impact than other sleep drugs…but that may not apply, though, to ME/CFS patients- only time will tell 🙂
If you have ever gone days and weeks without sleep, you will do almost anything to achieve it. Trying everything that we can to achieve sleep is the foundation for all of our bodies. Sleep is one of the most basic needs that we have. I almost lost my mind and felt like I was dying when I struggled so severely with sleep issues. Sleep has been an issue for me since day 1 with this illness. Public Citizen is not anyone’s friend as far as being a public watchdog. People with severe sleep issues need to be looked at differently than people who just occasionally can’t get a good night’s sleep. Drive? Can’t do it anyway due to sensory issues and being too tired for lack of sleep! It seems like everything we face is a catch 22.
I did wonder about people not being able to drive because they were too tired from lack of sleep! The placebo group, however, had less driving issues than the surovexant group but I think that was a small study; maybe they needed more hard-core insomniacs.
For me a couple of days of low sleep is so exhausting. Not being able to sleep seems to effect just about everything..I can’t imagine going for weeks on little sleep..although I know it happens frequently in ME/CFS.
I find research on sleep very conservative and strange. I have lots of problems with sleep, but is much better on a diet. Not only am I better without starches etc, but also if I avoid absolutely everything that gives an “up-feeling”: black pepper, lemon grass, tea, chocolate,…
Wake in the middle of the night: low cortisol.
Helps with rest, diet and vitamin B5.
Etc.
As long as researchers don´t look at diet and inflammation markers etc we will come nowhere, just expencive pills with side-effects.
If you use google translate I suppose you can understand my blog, most of the references are in English anyway.
Totally agree with this post. Please-I hope everyone doesn’t think I am coming down on them for taking meds I am not. However there are a lot of things you can do without an Rx or Dr. to improve your sleep and your health.
This is what helps me- Found a good sleep specialist after my first one-(don’t laugh) fell asleep while talking to me in a conference.
New sleep specialist retitrated my CPAP machine to a lower pressure. I also started when I would wake up after 3 hours or so-doing stretching exercises in the bed to increase circulation. I go back to sleep within 10 min.Sometimes if I am stressed-I take a 3 mg Melatonin or 5 HTP.
I am on a Gluten free diet. Very easy these days. NO Lactose.
Also I do not eat after 7:00 PM other than high alkiline water or an apple.
Anything spicy will keep you awake!!!Sometimes I will have an Electrolyte drink.
PROBIOTIC-VSL#3- 1 time a day. The Schiff Probiotic 2-4x day.
I do take a Mag creme rub on the back of my legs that I get from Our Kids and it relaxes my muscles (small amount).
Many other vitamins. I would say the CPAP has helped me the most. I actually after having Heart Palpitations and Racing heart decided myself to ask about the CPAP after someone on a website said it helped with heart symptoms.
All of my Palpitations are gone and my racing heart is gone.I still have the same stress I had before.Actually had been diagnosed with Heart Failure
by a well known CFIDS Dr.
Please consider holding off waiting for the next big drug and do some of these things for yourself!!! You may be surprised.
Carole
Sounds like good news for all of us who suffer from not getting enough sleep and not getting a sound sleep. I am all in favor of trying this drug. People who do not have sleep problems cannot begin to understand what it is like to not get the sleep we must have. So, bring this drug on and let us decide if it is something that benefits us. Especially in the CFS population, we are not out driving around, going to work, etc. We just need sleep. Thanks for the info. on this. Keep us posted..
The pundits are pretty sure it will get approved – probably at the lower doses. The FDA is expected to make a decision in 3rd/4th quarters of this year, which means it could happen any time.
My guess is that insurance companies will cover it and you won’t have to worry about the ME/CFS tag; all you need to do is have insomnia (???)
I wonder if it will get prescribed for non-insomniacs who waken early? It does appear to increase sleep efficiency; ie reduce sleep awakenings.
Another issue is price…..no ideas on that…
It’ll be interesting to see how it works out in ME/CFS. As Issie noted you don’t know until you know 🙂
Just one thing of question —sleeping pills affect GABA – GABA is what helps the parasympathetic system to turn on. Glutamate is the excitory one, that would keep one alert and awake. We want the GABA receptors to kick in and work. How does this drug affect sleep and inhibit GABA? I’m confused on this point.
Here’s an article on the drug you are talking about —looks like research at this point was on monkeys and rats. Appears it may not affect the memory like traditional sleep meds do. That would be good – if that’s true, it may not make for more grogginess in the daytime for CFS people. But, like Cort mentioned – if someone doesn’t eliminate meds properly due to liver dysfunction – it may linger longer and therefore not be a good thing. .
http://news.sciencemag.org/sciencenow/2013/04/a-sleep-drug-without-the-side-ef.html
Here’s another article showing this drug can cause dizziness, vivid dreams and memory problems – now that it’s in Phase 2 of trials. It says the main ingredient is doxepin. Which by the way is used a lot with mast cell people because it is an antihistamine and does help sleep. I however, could not take it —made me feel awful and had a paradox reaction that ramped up my system.
http://online.wsj.com/article/SB10001424052702304567604576454102061138630.html
There are lots of chemicals involved with sleep, an imbalance in any of these could cause potential problems. I found this article especially interesting.
http://www.sleepdex.org/neuro.htm
Issie
The main ingredient is doxepin! Ha…Dr. Cheney uses doxepin elixir and I think other ME/CFS experts probably do as well. …I think I remember Cheney saying something about it affecting histamine…fascinating stuff (who knew?)
I use doxepin, for 12 years now, works great, thank you Dr. Cheney!
Sorry- Don’t agree with you on anything on Cheney. He is the one that told me I was in Heart Failure. I saw him for 10 years. Gave up doing anything-as that is what he said I had to do. Never been sicker than on his regimen.Took me 18 mo to start getting better. Off of all of his treatments.
Every one does not have Heart Problems.!!!!!!
Yes, that paragraph about other sleep drugs working by “turning off GABA receptors” is incorrect. However that this new drug leaves GABA receptors alone is the point worth making.
Issie- You are so RIGHT!!!!!!
thanks cort for an interesting read. i am looking forward to this new drug. joanierav
Cort, speaking of Dr. Cheney and doxepin. Ha! is right, lol! That was a winner for me. For the 15 years I saw Dr. Cheney he had me taking a micro dose of doxepin. He prescribed an elixir of only about 5 mg (an uber low dose) it knocked me out with no side affects. He used it because he and other CFS doctors thought it had an immune modulating effect. Feel it did. Great heads up on this new drug. Looking forward to it, particularly because the main ingredient is doxepin, and it’s impact on histamine.
Good reporting Cort.
Best,
Questus
What is a doxepin elixir? I started taking doxepin with instructions to take as many as 10 mg a night I needed to knock me out. I couldn’t go to sleep but once I was asleep I couldn’t wake up…I’d try at 7, then 11 and finally around 1 in the afternoon I could get dressed. Of course, I broke doctors’ orders and reduced it and finally gave it up.
Are you saying we only need a small amount of doxepin per Cheney?
Doxepin is related to tricyclic antidepressants. IT may help SOME people, but people sensitive to histamines and tricyclics (especially people with Restless Legs Syndrome) cannot take this at all. If any of you have RLS do your research carefully. And not all people with sleep issues have sleep apnea. I am glad CPAP helps some of you, but lots with Fibro also have RLS. NOT a good fit at all for us. Just a friendly warning.
Really happy to see that, as my grandmother also have the same disorder. Apart from these drugs and medical treatments, I have also heard about the energy transmissions. Many people are tackling these problems through alternative friendly therapies/techniques that have a positive impact on health and well being. One such technique is receiving Energy transmissions. These can only be transmitted by only a few spiritual leaders on the planet. I couldn’t believe that these things are possible, but when i read peoples responses and there own experiences, I was thrilled!! It really changed many peoples life. Really Amazing!!
It’s not your everyday way of healing but thanks for sharing your experience. 🙂
The FDA has all the information they need to approve the lower doses of suvorexant, so what is the delay? I would like their explanation of wha they are accomplishing by prolonging the availability date. I fear it;s something like “good enough for government work”. Answers anyone?
I’ve been waiting almost a year for this to come out. Nothing helps me! Melatonin only regulates ones natural sleep pattern , which, I don’t have. I have severe delayed sleep phase syndrome & barely get into the first phase of sleep. That was when I was taking choral hydrate, which the pharmaceutical company discontinued 8 months ago. I really haven’t had any sleep in 8 long months! Which has lead to several nervous breakdowns. I’m just waiting each day for the call on this, that it has been approved by the FDA. I’ve called Merk several times & they say to just keep calling back. I wanted to be in their study, but it was already completed a year or 2 ago. I’m desperate for my sleep, so I can get a part time job & be able to function in this life again. This is literally killing me! I’m surprised I am not dead yet, from my lack of sleep & my heart condition. This is vital for me to try & I am having high hopes for Suvorexant. Any day now? I sure hope so!
No sleep will definitely do that to you. I hope this works for you 🙂 Good luck!
Do we know which countries this drug will be available in?
Where can I find this Dr. Cheney